KYAMCH-BRS Clinical Trials
Detailed project overview and case study

Overview
The Clinical Trial Management System (CTMS) for Khwaja Yunus Ali Bioresearch Services (KYABRS) is a digital platform designed to streamline the entire lifecycle of clinical trials. It supports researchers, administrators, and clinical teams in organizing, tracking, and monitoring all aspects of volunteer-based medical trials. The system enhances transparency, accuracy, and efficiency from planning and participant recruitment to data collection and reporting. KYABRS, as one of the oldest and most respected contract research organizations in Bangladesh, specializes in Phase I–III clinical trials, bioequivalence studies, and operates advanced facilities with dedicated screening areas, 106 volunteer beds, and over 40,000 sq. ft. of clinical trial space.
Key Features
- Centralized database for volunteer profiles
- Tracking participant recruitment, consent, and engagement
- Configurable workflows for different trial phases
- Secure storage of trial data and documentation
- Multi-study management
- Role-based access control for data security
- Exportable reports for internal review or regulatory use
Challenges & Solutions
One of the main challenges was ensuring sensitive trial data remained secure and error-free. Clinical trials involve personal and medical information, so we implemented secure login systems, encrypted data storage, and audit trails to maintain data accuracy and compliance. Another challenge was generating detailed reports quickly; we solved this by integrating pre-built report templates and export tools. Adoption was also a concern, so we designed a clean, intuitive interface and provided training materials and sessions for smooth onboarding.
Gallery

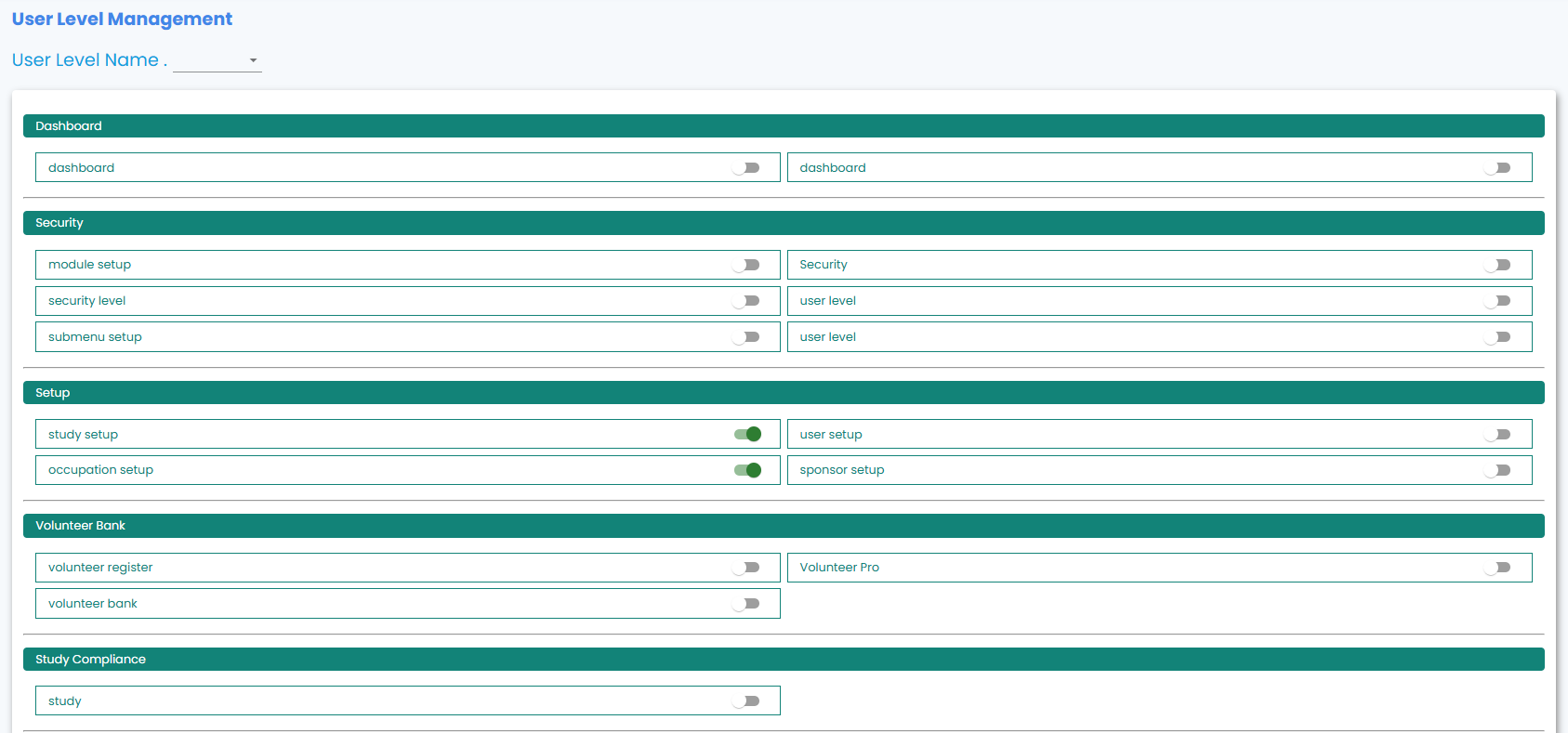
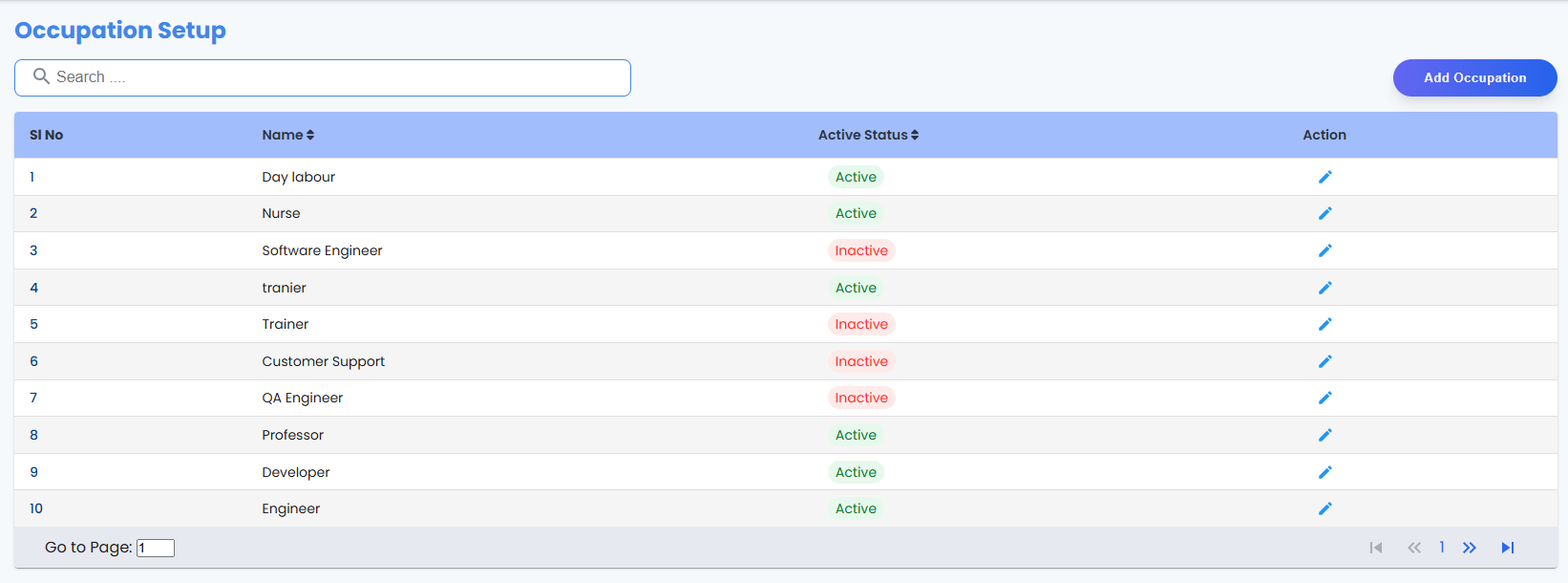
Project Details
Completion Date
2022
Project Duration
9 months